Clinical trials are crucial in driving medical innovation, yet many people find the process confusing or intimidating. By shedding light on how clinical trials work and the steps involved, patients and caregivers can better understand the opportunities to participate and the value of their contributions to the future of healthcare. If you are curious about the different clinical trial phases, you are not alone. Learning about these stages helps individuals make well-informed decisions about their health and the advancement of science.
Many significant treatments and medical breakthroughs rely on volunteers’ contributions to clinical trials. However, it is natural to have concerns about safety and the unknowns of emerging therapies. Knowing the structure and ethical safeguards of clinical research can dispel some of these worries, making participation a more approachable option. This article aims to provide a clear overview of clinical trials for anyone considering their options or simply wanting to understand how modern medicine progresses from the laboratory to widespread patient care. Aside from the personal benefits, participating in clinical trials means being a part of a global effort to bring more effective treatments to people in need. Each phase and step in the process is designed to ensure safety and generate useful knowledge. Experts, including medical professionals and regulatory bodies, oversee the journey at every step. Because misconceptions about clinical trials persist, it is vital to equip yourself with accurate information. The following sections will address the phases, participant criteria, ethics, benefits and risks, and resources for finding appropriate trials.
What Are Clinical Trials?
Clinical trials are carefully designed research studies that involve human volunteers, aiming to assess the safety and effectiveness of medical treatments, drugs, devices, or interventions. They bridge the gap between laboratory research and real-world medical practice. Before a treatment can be approved for use by the general public, it must undergo this systematic evaluation to meet strict scientific and regulatory standards. By participating in these studies, individuals contribute directly to the development and approval of new therapies for conditions with few or no existing options. The data gathered in clinical trials helps both researchers and regulatory agencies determine if a treatment works, for whom it works best, and whether its benefits outweigh potential risks
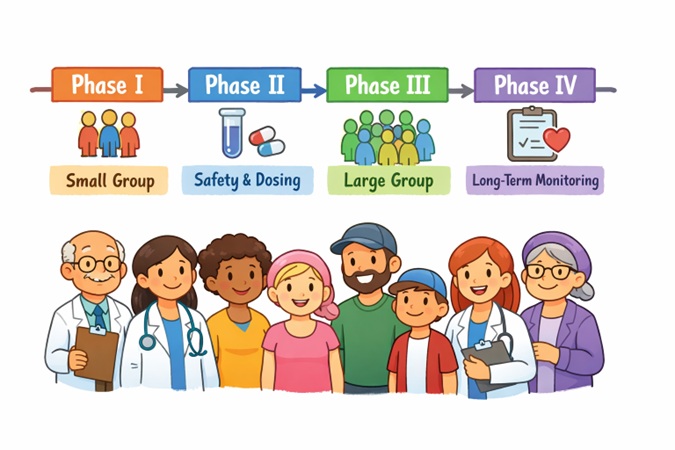
Phases Of Clinical Trials
Clinical trials are conducted in four distinct phases, each progressively involving more participants and collecting new types of data.
- Phase I: This phase involves a small group of healthy volunteers, focusing primarily on safety, appropriate dosage, and identifying possible side effects.
- Phase II: The trial expands to involve more people, often individuals who have the targeted condition, to study effectiveness and further evaluate safety.
- Phase III: Larger groups, sometimes thousands of participants across multiple sites, help confirm the treatment’s effectiveness, monitor side effects in a broader population, and compare it against existing treatments.
- Phase IV: Conducted after regulatory approval, this phase gathers additional information about the treatment’s risks, benefits, and optimal use over longer periods and in diverse patient groups.
Each phase adds crucial data, improving the understanding of a new treatment and guiding regulators as they determine whether to bring it to market.
Who Can Participate?
Participant eligibility for clinical trials is guided by clearly defined criteria that may include age, gender, medical history, and specific conditions or disease stages. These criteria, outlined in the study protocol, help ensure participant safety and the integrity of the research. Some studies require healthy volunteers, while others are designed for patients with specific diseases or medical profiles.
Researchers screen potential volunteers for both inclusion and exclusion criteria. Inclusion criteria define who is eligible for the study, while exclusion criteria identify conditions or factors that would make participation unsafe or that could interfere with study results. Consulting with your healthcare team about your eligibility and discussing any questions with the study coordinator is always encouraged.
Safeguards & Ethical Considerations
Strict ethical standards govern every clinical trial to prioritize participant welfare and transparency. Institutional Review Boards (IRBs) review study plans and informed consent documents before any participant can be enrolled. Informed consent means that all participants must receive and understand details about the study, including its purpose, procedures, risks, benefits, and their rights, before agreeing to take part. The rights of clinical trial participants are protected by national and international guidelines, such as the Declaration of Helsinki and Good Clinical Practice standards. Participants maintain the right to withdraw from a study at any time, for any reason, without penalty. Updates and continuous monitoring ensure that trials stay ethical and safe as they progress.
Benefits & Risks
Joining a clinical trial may offer several advantages, such as early access to cutting-edge treatments, close monitoring from a dedicated healthcare team, and the satisfaction of contributing to science and future patient care. However, it is essential to consider potential risks, including side effects, adverse reactions, and the possibility that the new treatment may not work as expected. Volunteers should ask questions, weigh potential benefits against known and unknown risks, and make decisions in discussion with their physicians. The process of evaluating risks and benefits is a key element of informed consent, empowering participants to make choices that align with their values and health goals.
How To Find Clinical Trials
Those interested in taking part can find active clinical trial listings on government databases. These databases provide detailed information about the purpose, eligibility, location, and contact information for each study. Speaking to your doctor or specialist is another valuable way to learn about studies that may be a good match for your medical history and needs. Patient advocacy groups and hospital research centers can also support the identification of opportunities.
Common Misconceptions
Widespread myths can discourage people from considering clinical trials. One common misconception is that they are only a last resort when standard treatments have failed. In reality, trials are often available at all stages of treatment for many conditions. Another myth is that participants are used as “lab rats,” but the truth is that rigorous protocols and oversight protect volunteers and ensure dignity and respect throughout the process. Understanding the facts, such as the thorough ethical review and continuous safety monitoring each trial receives, can help dispel these fears and promote a better experience for all participants.
Conclusion
Clinical trials play an essential role in developing new medical treatments and improving patient care. By understanding the process, phases, eligibility, and ethical safeguards, individuals can confidently consider participation. Well-informed volunteers are the foundation of successful research, contributing to breakthroughs that have the potential to transform healthcare for generations to come.
